
For Michael Konrad of South 40 Pier, science is personal. Here’s a memoir about his student days at the dawn of the nuclear era:
As people age, they often think about the past, their roots. Finding Your Roots is a popular PBS television show in which Henry Louis Gates Jr. and his staff research the history of guests invited to their show. Drama is created when these guests are presented with a deep history previously unknown to them.
I had a similar eerie experience two years ago when reading a chapter in an astronomy book on the creation of the chemical elements. The book mentioned that in 1953 Ward Whaling had discovered how carbon was created in super novae. Ward Whaling is a distinctive name, so I remembered that I knew him many years ago, 70 to be exact. I had just enrolled in Caltech as a physics major and was impatient with my professors’ insistence that I needed years of math and classical physics studies to even start to understand the research they were doing. I wanted to get into a lab, twist knobs, read dials, do nuclear physics!
Ward Whaling was a young professor of physics at Caltech and the Resident Associate for my dormitory. His job was to keep the boys out of trouble, at least serious trouble (my class in 1954 was about 125 men; in 2025 the freshman class was 248, 45 percent women). The dean told the new Associates each year, “if the boys do something bad, I want to hear about it from you before the police chief”. I asked Ward if it would be possible just to be an observer in his lab. He said he needed to think about it. Next week he told me that I could visit for a few hours each week if I promised not to ask too many questions or get in the way of real work. He explained they were using a van de Graaff generator [Fig. 1] to smash nuclei together.

In a van de Graaff generator static electrical charge is applied to a moving belt and transferred to the inside of a hollow metal dome which can attain 100 thousand volts. The generator Whaling used was about 20 feet tall and could reach 5 million volts. The toy version shown above is two feet tall.
I had a great time for a few months before I became bored and found I needed more time for homework. I was even able to help them once when they had a problem with an amplifier. The problem was noise, and because I was a radio amateur I guessed that replacing a glass vacuum tube with one covered with a metal shield might help. I rushed back to my room, found one in my junk box, and ran back to plug it in. It worked and I was on top of the world for days. But I didn’t really know what the lab was doing and how it fit into the physics of the day. Now I discover that Ward’s lab was actually doing important work.
The Caltech autobiographies
Fortunately, a Caltech tradition enabled me to learn what this work was. Retiring Caltech staff are interviewed about their work and life at Caltech, creating professional autobiographies, now available on the Internet. Whaling spent his entire career at Caltech and the head of Kellogg Lab, William Fowler, spent most of his career at Caltech also. Thus, 70 years later I was able to learn the details of the work Whaling’s lab was doing and what he thought about the students also [1, 2].
Hydrogen stars
Most of the mass in the universe is stars, planets represent only a tiny fraction. If you had asked any astronomer in the early 1900’s what elements the stars were made of they would have said their composition was similar to the earth’s; why wouldn’t they be? However, in 1925 a young woman graduate student at Harvard (one of the first), Cecilia Payne-Gaposchkin, described the spectra of stars in her Ph. D. thesis [3]. The spectra showed that stars are mostly hydrogen and a small amount of helium.
Hubble’s expanding hydrogen universe
A few years later, in 1929, Edwin Hubble was using the 100 inch Hooker telescope, the largest in the world at that time, at the top of Mt. Wilson, (only 10 miles from Caltech). The observatory had been founded by George Hale 20 years earlier, who realized that to do the best astronomy you not only need the best telescope, but also a location with good “seeing”. The telescope Payne-Gaposchkin had to use was in Boston, near the Harvard campus of course. There couldn’t be a much less suitable location. On the other hand, at that time Los Angeles was a modest size, the sky was clear most of the year, and the 5,000 foot high mountain range created exceptionally stable air and was above most of the modest haze and night light over the central part of Los Angeles.
Hubble discovered two extremely important facts about the universe. First, his large telescope and the clear air enabled him to see that the many smudges in earlier photographs made using smaller telescopes were actually galaxies, like the Milky Way Galaxy we live in. Thus, the universe was immensely larger than commonly thought at the time. Secondly, spectra of these distant stars showed that they were receding from one location in the universe at a speed proportional to the distance from that point. When you extrapolated positions back in time they must all have all come from that point. This extrapolation gave the first estimate for the age of the universe, about 15 billion years. The very early, tiny, compressed universe must have been so hot that matter could not exist, the universe was all energy. The extremely rapid expansion of this energy, which created the universe as we see it now, has been nicknamed the “Big Bang”.
As the energy radiated outward during the big bang it red shifted [4], the temperature dropped, protons and neutrons formed and then became hydrogen and helium nuclei. The heavier elements are just larger clumps of protons and neutrons, but the expansion was so rapid that essentially none were formed. This was the early hydrogen universe.
Stars are born and die
The hydrogen was a very dilute, fairly uniform, gas cloud. However, over hundreds of million years very small differences in the original amount of hydrogen in different locations became clumps due to gravitational attraction between hydrogen in the clumps. These clumps became large and massive as they accumulated more hydrogen and became very hot due to compression. When the temperature reached several million degrees the hydrogen nuclei began to fuse, making helium and releasing a huge amount of energy which increased the temperature even more. A star is born! Its aging will depend on its initial size. Let’s follow the life of a medium size star.
Our new star is almost in equilibrium. Radiation pressure from the hot center keeps the cooler outer shell of the star from collapsing, it’s now stable. As an example, the temperature at the center of our sun, a fairly new star, is several million degrees, while the surface is “only” 7,000 degrees. But maintaining this high temperature requires slow conversion of hydrogen to helium; the star is eating itself! After several billion years the star “burns” its hydrogen (not burning in the normal sense of course), then helium, then lithium, beryllium, etc. Not only is the star burning itself, but the resulting heavier elements produce less and less energy as they in turn burn. Iron is the end of the line, adding anything to iron requires energy, it doesn’t produce energy. This is not going to end well.
If the original star was above a certain size the gravitational force will become so large that its core then collapses, releasing a huge amount of energy and becoming the brightest object in its galaxy for several days or even months: it’s a supernova. The decay of a supernova is slow and hot enough that there are both the time and energy to make all the heavy elements, even those heavier than iron.
Ivy Mike
Astronomers and physicists began to make theories about the many nuclear fusion reactions that must occur inside stars and super nova even before the first atomic bomb was tested in 1945 [4]. Afterward, both theoretical and experimental work accelerated in the effort to develop a hydrogen, or fusion bomb, the “Super”. The first full test of a fusion bomb, in 1952, was named Ivy Mike. The test was heavily instrumented, and the results confirmed much of the theory on the nuclear reactions occurring in stars.
Stumped at carbon
Thus, by early 1953 plausible routes for nucleosynthesis of most of the elements had been proposed and some reactions had been confirmed experimentally. However, no one could propose a plausible mechanism for the creation of carbon, and carbon seemed a required intermediate for synthesis of the next heavier elements.
The carbon atom is also very important for us. It can form 4 strong bonds and thus generate the many complex molecules that are the basis of life [5]. The branch of chemistry known as organic chemistry is basically the chemistry of carbon containing molecules.
The carbon nuclear problem was that the total energy of likely precursors was much larger than the energy of the carbon nucleus itself. One of the rules for nuclear reactions is that the total energy of the precursors must be close to the total energy of the products.
Fred Hoyle at Cambridge, England had the confidence to predict that an unknown and very transient high energy state of Carbon must exist, and he calculated what its energy must be. This unknown state of carbon must be so unstable that it decays quickly by emitting a gamma ray and thus had not been observed.
He4 + Be8 -> C12* -> C12 + 𝛾 @ 7.367 MeV C12* = the transient, Hoyle carbon
There was only one place where experiments to prove this theory could be done quickly, and that was the Kellogg Lab at Caltech. Hoyle immediately wrote a letter to Fowler (people communicated by letters in those days) and to demonstrate his conviction traveled to Caltech to convince Fowler and give lectures on his ideas to anyone who would listen.
Whaling was the youngest professor in the Kellogg Lab. With a little prodding from Fowler, he agreed to look for the Hoyle carbon. It wouldn’t take much time, they did these kinds of experiments all the time, and he would get more experience with the equipment even if he didn’t find the Hoyle carbon. But he did find it and presented the results at a meeting that summer. That was what Whaling’s group had been doing the summer before I visited his lab. Whaling got tenure and Fowler got a Nobel prize! To be fair Fowler’s prize was for a large body of important work, not just discovery of the Hoyle carbon.
In Figure 2 (below), He4 (helium 4) is an alpha particle and Be8 (beryllium 8) is two, so the reaction is sometimes called the triple alpha reaction.
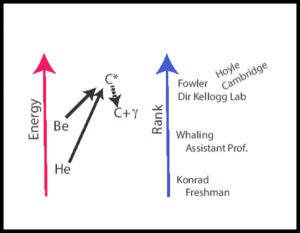
What Whaling thought of Caltech undergraduates
He was surprised at how rowdy we were. Perhaps that was the result of it being an all-male school then. Women can have a civilizing influence at that age.
References
-
-
-
-
-
- Ward Whaling Oral History Interview, interviewed by Charlotte E. (Shelley) Erwin,Caltech Archives Oral History Project, April 6, 1999, May 14, 1999,
http://resolver.caltech.edu/CaltechOH:OH_Whaling_W. - William Alfred Fowler Oral History Interview, interviewed by John L. Greenberg, Carol Bugé,Caltech Archives Oral History Project, May 13, 1983, May 31, 1984, October 3, 1986,
http://resolver.caltech.edu/CaltechOH:OH_Fowler_W. - What stars are made of; the life of Cecilia Payne-Gaposchkin.
by Donovan Moore, Harvard University Press, 2020 - The Greenhouse Effect by Michael Konrad, Floating Times, 2025.
https://ft.floatinghomes.org/the-greenhouse-effect/ - Diatoms: algae that live in glass houses by Michael Konrad, Floating Times, 2024. https://ft.floatinghomes.org/diatoms-algae-that-live-in-glass-houses/
- Ward Whaling Oral History Interview, interviewed by Charlotte E. (Shelley) Erwin,Caltech Archives Oral History Project, April 6, 1999, May 14, 1999,
-
-
-
-